SAFITRI, LARAS INTAN (2015) PENURUNAN KADAR BESI (Fe) DENGAN METODE ANALISA PERMANGANOMETRI DALAM ION EXCHANGER PADA AIR SUNGAI UNDIP (Decreased Content of Iron (Fe) Using Ion Exchanger with Permanganometri Analysis Method in Diponegoro University’s River Water). Undergraduate thesis, Undip.
| PDF (COVER, HALAMAN PENGESAHAN) - Published Version 138Kb | |
| PDF (KATA PENGATAR) - Published Version 144Kb | |
| PDF (DAFTAR ISI) - Published Version 95Kb | |
| PDF (BAB I) - Published Version 86Kb | |
| PDF (BAB II) - Published Version 179Kb | |
| PDF (BAB III) - Published Version 82Kb | |
| PDF (BAB IV) - Published Version 369Kb | |
| PDF (BAB V) - Published Version Restricted to Repository staff only 120Kb | ||
| PDF (BAB VI) - Published Version Restricted to Repository staff only 336Kb | ||
| PDF (BAB VII) - Published Version Restricted to Repository staff only 84Kb | ||
| PDF (DAFTAR PUSTAKA) - Published Version 142Kb | |
| PDF (LAMPIRAN) - Published Version 342Kb |
Abstract
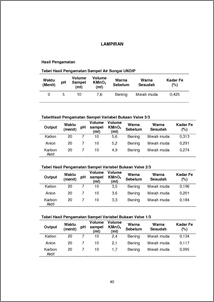
According to Erlinda (2014), iron is one of the elements that can be found in almost any place on earth, in all of the geological layers and all bodies of water. In general, the iron in the water can be dissolved as a salt compound ferric (Fe 3+) or ferrous salt (Fe 2+) were suspended as colloidal grains (diameter <1) and incorporated with organic substances or inorganic solid. Substances that are absorbed by the water can be classified as dissolved solids, dissolved gases and suspended solids. In general, the water contained in inorganic substances such as iron (Fe), manganese (Mn), zinc (Zn), lead (Pb), and so on. Fe is an essential heavy metals, which in some measure is needed by the human body, but excessive levels may cause toxic effects. Ion exchange resin or ion exchange resin can be defined as polymerized hydrocarbon compounds, containing crosslinking (crosslinking) as well as functional groups that have ions that can be exchanged. The types of resin that is strong acid cation resin, weak acid cation, anion strong base and strong base anion. The principle of ion exchanger is in the process of insoluble compounds, in this case the resin, received a positive or negative ions from solution and release certain other ions into the solution in an amount equivalent to the same. If the ions are exchanged in the form of a cation, then the resin is called cation exchange resin, and if ions are exchanged in the form of anion, the resin is called an anion exchange resin. The series of ion exchangers tool consists of three tanks, the tank cation exchanger, anion exchanger tank, and activated carbon tanks, each of which is made of FRP. At the top of the tank there is a way valve to the filter, fast rinse and back wash. The method used in the analysis kadarFe is permanganometry the process of titration for determining the concentration of a reducing agent using an oxidant (KMnO4) as a standard solution. In this lab to test the Fe content is done by means of Ion Exchangers, with as much as 100 liters of water each variable. The variables used are the valve 1/3, 2/3, and 3/3. Time used for 20 minutes and take 10 ml of the sample in the tank cation, anion active and more carbon. Results of observations obtained tank Fe content of cations, anions, activated carbon of 0.134%; 0.117%; 0.095%. At 2/3 variable of 0.196%; 0,201%; 0.184%. And the 3/3 variable of 0.313%; 0.291%; 0.274%. It can be concluded that the greatest reduction in Fe content occurs at the smallest aperture or variable valve 1/3 (Keywords : Levels of Fe, Ion Exchangers, resin)
| Item Type: | Thesis (Undergraduate) |
|---|---|
| Subjects: | T Technology > TP Chemical technology |
| Divisions: | Faculty of Engineering > Diploma in Chemical Engineering Faculty of Engineering > Diploma in Chemical Engineering |
| ID Code: | 48048 |
| Deposited By: | INVALID USER |
| Deposited On: | 18 Mar 2016 09:58 |
| Last Modified: | 18 Mar 2016 09:58 |
Repository Staff Only: item control page